
FAQs
We are hoping to raise awareness of RVCL and symptoms to support our mission of supporting meaningful research to find treatments and a cure.
Retinal Vasculopathy with Cerebral Leukoencephalopathy
RVCL is a rare genetic disease that affects the small blood vessels. Inherited mutations in the TREX1 gene cause RVCL. RVCL is associated with damage to multiple organs, including the brain, eyes, kidneys, liver, and bones. This leads to disability, vision loss, and premature death in all cases, but we are working to find effective treatments and develop personalized medicines that target TREX1. There are fewer than 200 known patients and 50 families with RVCL in the world.
Click the menus below to learn more about RVCL, including genetics, diagnosis, symptoms, and treatments.
-
RVCL or RVCL-S stands for Retinal Vasculopathy with Cerebral Leukoencephalopathy (and Systemic manifestations), also known as retinal vasculopathy with cerebral leukodystrophy, cerebroretinal vasculopathy (CRV), or hereditary endotheliopathy with retinopathy, nephropathy, and stroke (HERNS). Other abbreviations also have been used, including CHARIOT and HVR.
The name of the disease is explained as follows:
Retinal = relating to the retina or the portion of the eye that senses light and sends images to the brain
Vasculopathy = blood vessel disease
Cerebral = brain
Leukoencephalopathy = white matter diseaseSystemic manifestations = an all-encompassing term reflecting the fact that patients with RVCL also develop kidney, liver, and sometimes also gastrointestinal, thyroid, and bone disease. Thus, RVCL is not just a disease of the brain and the eye. It is truly a multi-system disease.
-
RVCL is caused by mutations in a gene called TREX1, which is found on chromosome 3. RVCL is inherited in an autosomal dominant pattern, which means that about half of family members are affected.
Each affected individual has a 50% chance of passing the disease to each child. Every person has two copies of the TREX1 gene, one copy inherited from each parent. Patients with RVCL have one normal copy of TREX1, and one mutated copy of TREX1. Only one copy of TREX1 is passed to the next generation, which is why there is a 50% chance of passing the disease to each child.
If you would like to find out about a procedure called pre-implantation genetic testing, you can read more about this on the “Finding a Cure” section of our web site. Pre-implantation genetic testing is a procedure that allows couples to perform in vitro fertilization and then choose embryos without the TREX1 mutation for transfer to the uterus.
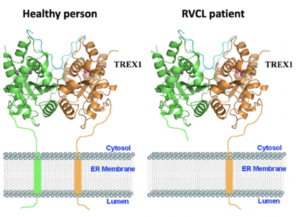
Healthy people have 2 full-length copies of TREX1 protein. Patients with RVCL have one mutated, short copy of the TREX1 protein and one full-length, normal copy of the TREX1 protein.
-
Each gene is the blueprint for a protein, and proteins are the machinery that make the human body work. The TREX1 gene encodes the TREX1 protein. Normally, the TREX1 protein is attached to a membrane inside the cell. In patients with RVCL, one copy of TREX1 is abnormally shortened because of the mutation. This causes the TREX1 protein to float freely in the cell, rather than being anchored to the membrane. Mislocalization of the TREX1 protein somehow causes cellular damage and disease. The exact mechanism remains to be defined, but therapies targeting the mutated TREX1 gene and protein are currently in development.
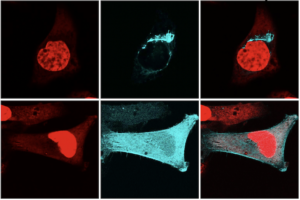
These are microscope images that show a single layer (a slice) through the middle of two cells. The upper cell has normal, full-length TREX1. The lower cell has the RVCL mutant form of TREX1. In red is the nucleus, where the DNA is located. The Cyan color is fluorescently labeled TREX1 protein. The upper panels show the normal location of normal, full-length TREX1 protein, which is attached to the membrane of the ER. The lower panel shows the location of untethered mutant TREX1, which causes RVCL. The mutant form of the TREX1 protein is found thoughout the cell instead of at the ER membrane.
-
RVCL is diagnosed based on a specific set of TREX1 gene mutations and is conclusively established by genetic testing for the TREX1 gene mutation. The mutations that cause RVCL are in a specific region of the TREX1 gene. Other mutations in TREX1 can cause other diseases, but all the mutations that cause RVCL are within a specifically defined region. Testing is available, but patients are encouraged to consult with their primary physician and family before deciding to test for the TREX1 gene mutation. Genetic testing should only be performed under the direct guidance and support of expert physicians and genetic counselors, can be used to accurately diagnose RVCL.
Unfortunately, many patients with RVCL are not genetically tested, and those patients can end up being diagnosed as having multiple sclerosis, brain tumors, lupus, or vasculitis. Those diseases are much more common than RVCL. Since most physicians have not heard of RVCL, they often presume that patients with RVCL have one of these more common diseases.
We are hoping to raise awareness of RVCL so that more patients can receive the correct diagnosis. However, before genetic testing, it is important to consult with expert physicians and a genetic counselor with expertise in RVCL.
-
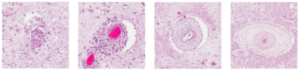
Typical blood vessel abnormalities, published in Kolar et al., Brain Pathology, 2014.
Early symptoms
Most patients with RVCL are generally healthy until around age 40-50. Occasionally, patients with RVCL develop symptoms earlier or later in life. Early symptoms can include Raynaud’s syndrome (fingers changing color in the cold), blind spots, or small strokes. Many patients develop fatigue, partial loss of vision, or memory problems. Other patients develop kidney, thyroid, or liver abnormalities earlier in the course of disease.Vasculopathy and organ damage in RVCL
One major process underlying RVCL is related to loss of small blood vessels. (Vasculopathy means “disease of the blood vessels.”) Loss of blood flow leads to progressive organ damage. Examples of organ damage in RVCL include chronic kidney disease, loss of blood flow to the retina leading to blindness or glaucoma, and strokes leading to neurological symptoms. Sometimes new symptoms can arise suddenly (e.g., glaucoma or strokes), but usually the disease is slowly progressive over the course of many years.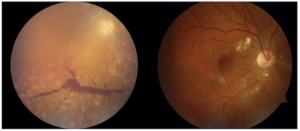
Patients with RVCL develop retinal eye disease (retinopathy). This can lead to vision loss. This includes areas of ischemia (lack of blood flow) due to loss of small blood vessels. Sometimes patients develop glaucoma, leading to sudden vision loss.
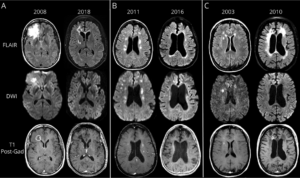
Typical progression of brain lesions in RVCL, published by Ford et al., Neurology, 2020.
Impact of RVCL on the brain
Brain imaging typically shows disease of the white matter, including large brain lesions in about 1/3rd of patients, and smaller brain lesions in about 2/3rds of patients. Patients with RVCL also develop brain atrophy, which was recently characterized in a study by our colleague, Dr. Andria Ford of Washington University. Progressive brain injury caused by RVCL is usually associated with memory problems or other neurological symptoms like weakness, numbness, or stroke-like symptoms.Physicians who monitor patients with RVCL typically perform eye exams with careful imaging of the retina, physical exams, cognitive testing, and brain imaging every 6 months, as well as blood work to monitor liver and kidney function every 3-6 months.
-
We are committed to supporting patients and families with RVCL. Clinical trials of Crizanlizumab for RVCL at the University of Pennsylvania and in collaboration with Washington University are underway. More information is available here.
Crizanlizumab targets a protein called P-selectin, which captures blood cells and can contribute to blockages in small blood vessels. Crizanlizumab is already FDA-approved for another disease associated with blockages in small blood vessels. You can read more about the research here.
-
Every insurance plan – even plans through the same insurance company – covers different expenses related to doctors, clinics, and other services. While we have seen great success with care being covered by various plans (e.g., Medicare Advantage Plan), you should always consult with your insurance provider. Some patients are surprised to learn that their plan covers more than they expected. Check that the care you want is covered.

